Disease relapse after allogeneic stem cell transplantation (allo-SCT) is the main challenge in achieving a cure for acute myeloid leukemias (AMLs) and myelodysplastic syndromes (MDSs). The treatment of disease relapse can be offered in case of full hematological recurrence or in the presence of minimal residual disease (MRD) positivity and/or mixed chimerism. The latter approach is defined as pre-emptive therapy. Overall, treatment strategies may include chemotherapy, molecular-target drugs, hypomethylating agents (HMAs), donor lymphocyte infusions (DLIs) or even a second allo-SCT.
We retrospectively analyzed a cohort of 553 AML and MDS adult patients consecutively allotransplanted between January 1, 2015, and December 31, 2021, at Saint-Antoine University Hospital, Paris (France) (n=420) and Spedali Civili di Brescia, Brescia (Italy) (n=133). 134/553 (24%) patients relapsed and were included in this study. 103/134 (77%) were subsequently treated. Among patients who received treatment, 40/103 (39%) were treated with a DLI-based regimen. In this group, 9 received DLIs alone, while the others also received HMAs (4 cases), HMAs+ venetoclax (12 cases), FLT3-inhibitors (3 cases), intensive chemotherapy (5 cases), a second allo-SCT (5 cases), or other therapies (2 cases). The remaining 63/103 (61%) patients were treated with regimens that did not include DLIs. Of these, 10 received HMAs, 27 HMAs+ venetoclax, 4 FLT3-inhibitors, 6 intensive chemotherapy, 9 a second allo-SCT, and 7 other therapies.
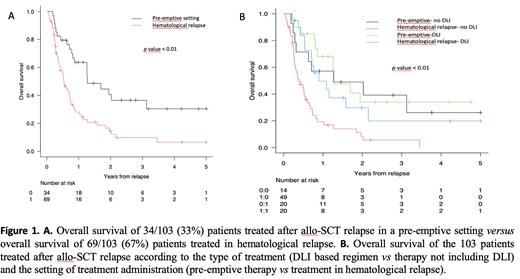
With a median follow up of 1.6 years, the estimated 1-, 2- and 5-year OS rate of the 134 patients who relapsed after allo-SCT was 32%, 18 %, and 11 %, respectively. The OS of patients treated after allo-SCT relapse was 40 %, 20%, and 15% at 1, 2 and 5 years compared with 6 %, 3%, and 0 % for patients who did not receive therapy, respectively ( p<0.01).Moreover, the OS was significantly better in patients treated in a pre-emptive setting than in those treated for a morphological relapse (OS at 1, 2 and 5 years was 60%, 36% and 30%, respectively, for patients treated in a pre-emptive setting versus 26%, 12%, and 6%, respectively, for patients treated in hematological relapse ( p<0.01) (Figure 1A). Focusing on post-relapse treatment, patients who received DLI-based regimens had a 1-, 2- and 5-year OS of 55%, 32% and 32%, respectively, compared to 27%, 16% and 7% for patients treated with other therapies ( p<0.01). Among patients who received DLIs, 50% of patients were treated in the pre-emptive setting. Conversely, in the group of patients treated without DLI, only 22% were treated in the pre-emptive setting, reflecting the more advanced phase of relapse in this group. Finally, when patients were analyzed by the treatment strategy (pre-emptive therapy vs therapy in hematological relapse) and DLI administration (yes vs no), the best outcome was achieved when the relapse was promptly detected and pre-emptive therapy was started, particularly if a DLI was administered. Indeed, the 1-year OS of patients treated in the pre-emptive setting was 67% for those receiving a DLI and 54% for those treated without DLI, while for patients with hematologic relapse, the 1-year OS was 43% for those treated with the DLI-based regimen and 17% for those treated without DLI ( p<0.01) (Figure 1B). On multivariate analysis, treatment with DLI after relapse, and pre-emptive setting were independent factors associated with better OS ( p=0.03 and p<0.01), respectively.
Our data showed that relapse treatment with pre-emptive therapies is associated with improved outcomes and this effect is more evident when combined with a DLI . These results confirm the primary role of the graft- versus leukemia effect in the treatment of relapse and underscore the importance of its early identification through MRD monitoring.
Disclosures
Malagola:Biotest, MSD: Consultancy, Honoraria. Mohty:JAZZ PHARMACEUTICALS: Honoraria, Research Funding. Polverelli:BMS: Honoraria; GSK: Honoraria; Abbvie: Honoraria; Novartis: Honoraria. Russo:Medac, Abbvie, MSD, Jazz Pharma, Gilead, Novartis: Membership on an entity's Board of Directors or advisory committees; MSD, Novartis, Gilead, BMS, Medac: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal